衣殼修飾的工程化AAV
1.研究背景
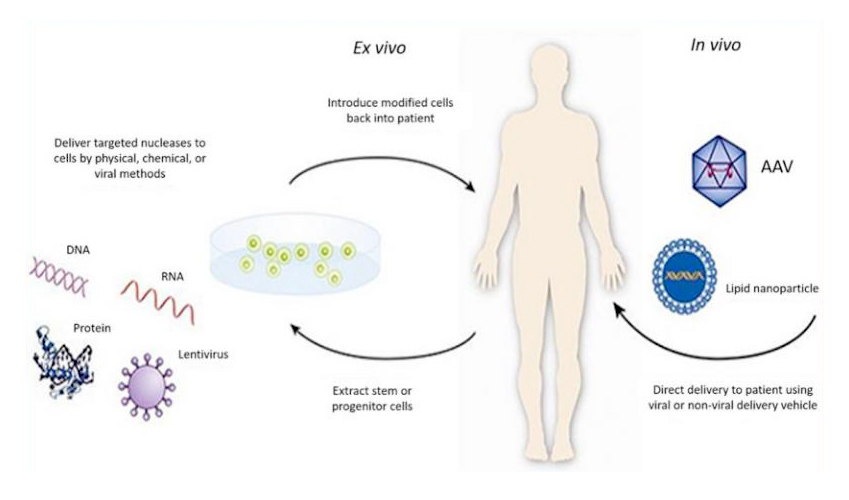
基因治療是一種新型的治療手段,它可以治療多種疾病�,包括癌癥�����、遺傳性疾病��、神經(jīng)性疾病��、感染性疾病、心血管疾病和自身免疫性疾病等�,它針對(duì)的是疾病的根源而不僅僅是疾病的癥狀。如此有效的治療方法有什么特殊性呢��?其實(shí)很簡單�����,它就是將外源正常基因借助一定的技術(shù)或者載體導(dǎo)入靶細(xì)胞�����,從而糾正或補(bǔ)償缺陷和異?��;蛞鸬募膊?���,以達(dá)到治療疾病的目的(圖1)��。
圖1:基因治療的示意圖(來源于:https://www.fda.gov/)
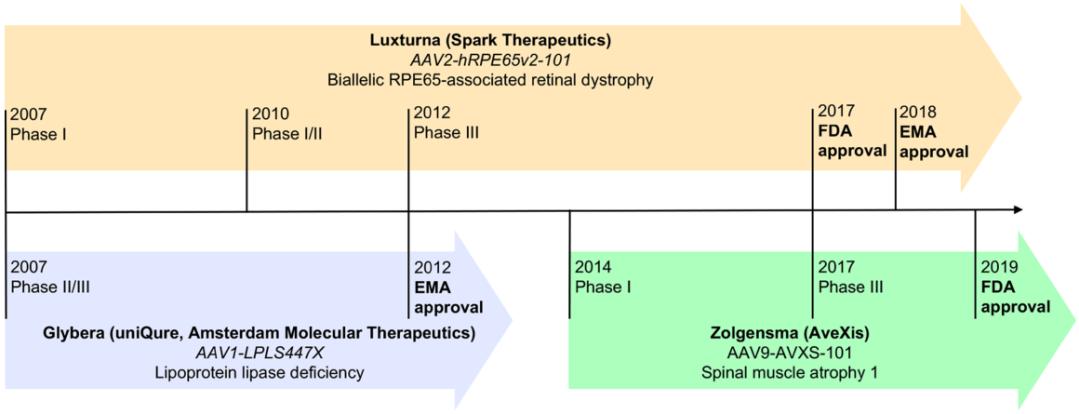
實(shí)現(xiàn)基因治療成功的關(guān)鍵是載體的選擇。在眾多載體工具中����,腺相關(guān)病毒載體(Adeno-Associated Viral Vector, AAV Vector)憑借其安全性高、免疫原性低和表達(dá)時(shí)程長的優(yōu)勢成為當(dāng)下基因治療中極具市場前景的病毒載體�����,從而被越來越多的臨床試驗(yàn)用于治療各種疾病�。截止目前�,已有3款基于AAV載體的基因藥物被獲批上市(圖2):第一款是2012年由歐洲藥品管理局(EMA)批準(zhǔn)的GLYBERA® (alipogene tiparvovec, AT)����,該藥物由荷蘭UniQure公司研制�����,用于治療脂蛋白脂酶缺乏癥(LPLD)����;第二款是2017年由美國食品藥品監(jiān)督管理局(FDA)批準(zhǔn)的LUXTURNA®(voretigene neparvovec-rzyl, VN)�����,次年又被EMA獲批����,該藥物由美國Spark Therapeutics公司研制�����,用于治療Leber先天性黑蒙癥II型�����;第3款是2019年同樣由FDA批準(zhǔn)的ZOLGENSMA® (onasemnogene abeparvovec-xioi, OA) ,該藥物由美國AveXis公司研制��,用于治療2歲以下患兒的脊髓性肌萎縮癥I型���。
圖2:3款獲批上市的基于AAV載體的基因藥物的時(shí)間表
(Lugin, M. L., et al. (2020). ACS Nano 14(11): 14262-14283.)
上述取得的成就��,再加上美國臨床試驗(yàn)數(shù)據(jù)庫(ClinicalTrials.gov)中的一系列臨床前和臨床研究均證實(shí)了AAV載體的安全性和有效性���。但是野生型AAV畢竟不是為了遞送疾病治療基因而進(jìn)化的。而且���,體內(nèi)預(yù)存的中和抗體和AAV載體的承載能力進(jìn)一步限制了AAV載體的廣泛應(yīng)用�����。目前�,科學(xué)家們研究的重點(diǎn)主要集中在工程化AAV載體�����,用于提高其安全性�、靶向性和轉(zhuǎn)導(dǎo)效率���。工程化AAV載體的構(gòu)建方法多種多樣�����,本篇我們將重點(diǎn)跟大家分享衣殼修飾的工程化AAV載體(圖3)�。
圖3:工程化AAV載體的示意圖
2.AAV基因組結(jié)構(gòu)
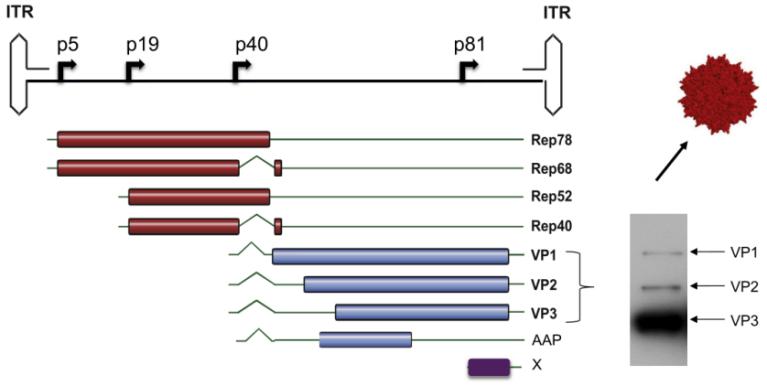
了解AAV基因組結(jié)構(gòu)是構(gòu)建工程化AAV的基礎(chǔ)�。野生型AAV含有約4.7kb的單鏈線性DNA基因組���,三個(gè)基因Rep, Cap和X的兩端各由145bp的ITR組成�����,可形成T型的發(fā)夾結(jié)構(gòu)(圖4)。Rep基因編碼病毒轉(zhuǎn)錄調(diào)控�、復(fù)制和包裝必需的四個(gè)調(diào)控蛋白:Rep78, Rep68, Rep52 和Rep40。Cap基因編碼三個(gè)重疊的結(jié)構(gòu)蛋白(VP1, VP2, VP3�����,其表達(dá)比例為1:1:10)和VPs入核必需的裝配激活蛋白AAP(assembly-activating protein)����。X基因編碼基因組復(fù)制中具有支持功能的蛋白質(zhì)��。
圖4:AAV2載體的基因組結(jié)構(gòu)
(Buning, H. and A. Srivastava (2019). Mol Ther Methods Clin Dev 12: 248-265.)
對(duì)于Cap基因��,我們從基因組的VPs基因分布上就可以看出,VP3被全部包含在VP2和VP1中�����,因此,VP3就是3個(gè)VPs的共同區(qū)域����,也被稱為VP3公共區(qū)��;VP2較VP3長約57個(gè)氨基酸的序列�,且該序列被包含在VP1中�,因此����,也叫VP1/VP2公共區(qū)����;只有VP1 N端獨(dú)有大約138個(gè)氨基酸的序列,因此�����,也叫VP1獨(dú)特區(qū)(VP1u)��。VP3公共區(qū)負(fù)責(zé)組裝二十面體衣殼;VP1u含有磷脂酶A2結(jié)構(gòu)域�����,VP1u和VP1/VP2公共區(qū)含有核定位序列NLSs�,因此�����,VP1u和VP1/VP2公共區(qū)負(fù)責(zé)內(nèi)體運(yùn)輸&逃離、核定位和基因組釋放���。
3.AAV衣殼結(jié)構(gòu)
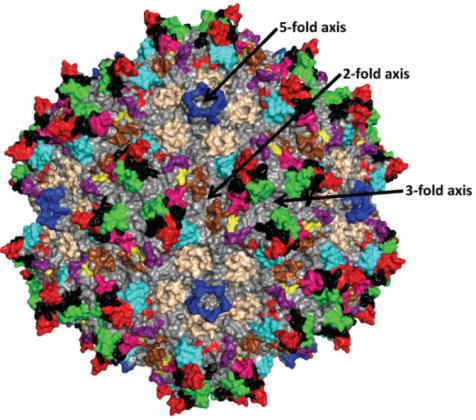
AAV衣殼結(jié)構(gòu)的解析有助于理解AAV如何感染細(xì)胞和確定工程化AAV衣殼修飾的位置。腺相關(guān)病毒的衣殼呈T=1二十面體(二十面體是由20個(gè)三角形圍成的凸多面體�����,每5個(gè)三角形圍出一個(gè)五倍頂���,通過每一對(duì)相對(duì)著的五倍頂有一個(gè)五倍旋轉(zhuǎn)軸;通過每一對(duì)相對(duì)著的三角形中心有1個(gè)三倍旋轉(zhuǎn)軸�;通過每一對(duì)相對(duì)著的棱的中點(diǎn)有1個(gè)二倍旋轉(zhuǎn)軸)��。它由60個(gè)VP單體通過這些旋轉(zhuǎn)軸的相互作用裝配而成,這些VP單體可以全部為VP3�,也可以由VP1����、VP2�、VP3共同構(gòu)成。
AAV衣殼表面的顯著特征是二倍凹地�,三倍凸起和五倍通道(圖5)����。圓柱形通道連通衣殼內(nèi)外,是AAV DNA進(jìn)入衣殼的地方�,它參與 Rep 蛋白結(jié)合��、衣殼蛋白裝配��、VP1 N 末端外露、AAV病毒感染等多個(gè)過程�;二倍凹地是病毒衣殼最薄的地方�����;三倍凸起的主要作用是識(shí)別受體��。
圖5:由60個(gè)VP3裝配成的AAV2衣殼的拓?fù)浣Y(jié)構(gòu)
(Tseng, Y. S. and M. Agbandje-McKenna (2014). Front Immunol 5: 9.)
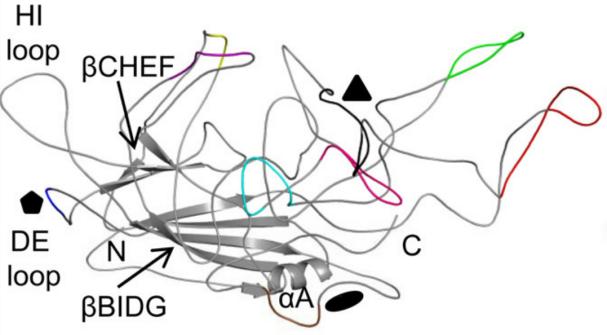
每個(gè)衣殼蛋白的拓?fù)浣Y(jié)構(gòu)由一個(gè)保守的α螺旋(αA)和一個(gè)由8股反向平行鏈(βB-βI)構(gòu)成的β桶核心組成(圖6)。在8股反向平行鏈之間存在4個(gè)大的鏈間環(huán)(BC���,DE,GH��,HI��,根據(jù)環(huán)所在位置命名)���,這些環(huán)構(gòu)成衣殼的外表面。其中��,GH環(huán)是較大的環(huán)�,又可以分為多個(gè)亞環(huán)��。這些亞環(huán)在主要的衣殼組分VP3中具有較高的序列和結(jié)構(gòu)可變性,經(jīng)鑒定包含9個(gè)可變區(qū)(variable regions; VRs)���,這些VRs在不同的血清間存在差異��,主要在受體附著����、組織轉(zhuǎn)導(dǎo)和抗原性方面起作用����。
圖6:AAV2 VP3單體的拓?fù)浣Y(jié)構(gòu)。9個(gè)VRs被用不同的顏色標(biāo)記:
I�,紫色�����;II���,藍(lán)色���;III,黃色���;IV,紅色����;V���,黑色�;VI���,粉紅色;VII�,青色��;VIII����,綠色����;IX���,巧克力色
(Gurda, B. L., et al. (2013). J Virol 87(16): 9111-9124.)
4.衣殼修飾的工程化AAV
特定的VP亞基已被作為衣殼修飾的靶點(diǎn)����,例如去除免疫原基序,整合標(biāo)簽或熒光基團(tuán)和重新靶向等�����。
AAV2是研究相對(duì)成熟的血清型����。以AAV2為骨架����,在公共VP3區(qū)����,大量研究表明�,I-587(VP1氨基酸數(shù))和I-588為較常用的衣殼修飾位置����,因?yàn)檫@2個(gè)位置地處三倍凸起附近����,可以接受長達(dá)34個(gè)氨基酸長度的肽鏈插入,而不影響衣殼化和基因組包裝�。而且����,外源肽鏈的插入使得AAV2第一受體結(jié)合基序被修飾,最終賦予AAV2變體新的靶向性�。Buning, H.等人匯總了在I-587和I-588處插入肽鏈的AAV2變體(表1)�����。維真生物現(xiàn)擁有表1所列的所有AAV Cap質(zhì)粒�,數(shù)量近70種�����,可直接進(jìn)行AAV病毒包裝。
包裝表1所列AAV血清型���,年中讓利僅需8折,還送免費(fèi)對(duì)照�!
我們承諾:包裝不成功不收費(fèi)���!
表1:在I-587和I-588處插入肽鏈的AAV2變體
|
Serotype
|
Position
|
Name
|
Target Cell Type
|
Insert
|
|
AAV2
|
I-587
|
AAV-I-587
|
β1-integrin positive tumor cells
|
QAGTFALRGDNPQG
|
|
AAV2
|
I-587
|
AAV-588NGR
|
CD13-positive tumor cells
|
NGRAHA
|
|
AAV2
|
I-587
|
AAV-MO7A
|
tumor cells
|
RGDAVGV
|
|
AAV2
|
I-587
|
AAV-MO7T
|
tumor cells
|
RGDTPTS
|
|
AAV2
|
I-587
|
AAV-MecA
|
tumor cells
|
GENQARS
|
|
AAV2
|
I-587
|
AAV-MecB
|
tumor cells
|
RSNAVVP
|
|
AAV2
|
I-587
|
rRGD587
|
αv integrin positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-587
|
AAV-C4
|
tumor cells
|
PRGTNGP
|
|
AAV2
|
I-587
|
AAV-D10
|
tumor cells
|
SRGATTT
|
|
AAV2
|
I-587
|
AAV-SIG
|
endothelial cells
|
SIGYPLP
|
|
AAV2
|
I-587
|
AAV-MTP
|
endothelial cells
|
MTPFPTSNEANL
|
|
AAV2
|
I-587
|
AAV-QPE
|
endothelial cells
|
QPEHSST
|
|
AAV2
|
I-587
|
AAV-VNT
|
endothelial cells
|
VNTANST
|
|
AAV2
|
I-587
|
AAV-CNH
|
endothelial cells
|
CNHRYMQMC
|
|
AAV2
|
I-587
|
AAV-CAP
|
endothelial cells
|
CAPGPSKSG
|
|
AAV2
|
I-587
|
AAV-EYH
|
smooth muscle cells
|
EYHHYNK
|
|
AAV2
|
I-587
|
AAV587MTP
|
skeleton muscle cells
|
ASSLNIA
|
|
AAV2
|
I-587
|
AAV-r3.45
|
neuronal stem cells
|
TQVGQKT
|
|
AAV2
|
I-587
|
AAV2-LSS
|
CNS
|
LPSSLQK
|
|
AAV2
|
I-587
|
AAV2-PPS
|
CNS
|
DSPAHPS
|
|
AAV2
|
I-587
|
AAV2-TLH
|
CNS
|
GWTLHNK
|
|
AAV2
|
I-587
|
AAV2-GMN
|
CNS
|
GMNAFRA
|
|
AAV2
|
I-587
|
AAV-Kera1
|
keratinocytes
|
RGDTATL
|
|
AAV2
|
I-587
|
AAV-Kera2
|
keratinocytes
|
PRGDLAP
|
|
AAV2
|
I-587
|
AAV-Kera3
|
keratinocytes
|
RGDQQSL
|
|
AAV2
|
I-587
|
AAV-588Myc
|
none
|
EQLSISEEDL
|
|
AAV2
|
I-587
|
AAV2.N587_R588insBAP
|
adaptor
|
GLNDIFEAQKIEWHE
|
|
AAV2
|
I-587
|
AAV2Ald13
|
adaptor
|
LCTPSRAALLTGR
|
|
AAV2
|
I-587
|
DMD4
|
vaccine
|
QVSHWVSGLAEGSFG
|
|
AAV2
|
I-587
|
DMD6
|
vaccine
|
LSHTSGRVEGSVSLL
|
|
AAV2
|
I-588
|
A588-RGD4C
|
av integrin-positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-588
|
A588-RGD4CGLS
|
av-integrin positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-588
|
AAV-VTAGRAP
|
tumor cells
|
VTAGRAP
|
|
AAV2
|
I-588
|
AAV-APVTRPA
|
tumor cells
|
APVTRPA
|
|
AAV2
|
I-588
|
AAV-DLSNLTR
|
tumor cells
|
DLSNLTR
|
|
AAV2
|
I-588
|
AAV-NQVGSWS
|
tumor cells
|
NQVGSWS
|
|
AAV2
|
I-588
|
AAV-EARVRPP
|
tumor cells
|
EARVRPP
|
|
AAV2
|
I-588
|
AAV-NSVSLYT
|
tumor cells (CML)
|
NSVSLYT
|
|
AAV2
|
I-588
|
AAV-LS1
|
tumor cells (CML), CD34+cells
|
NDVRSAN*
|
|
AAV2
|
I-588
|
AAV-LS2
|
tumor cells (CML), CD34+cells
|
NESRVLS
|
|
AAV2
|
I-588
|
AAV-LS3
|
tumor cells (CML), CD34+cells
|
NRTWEQQ
|
|
AAV2
|
I-588
|
AAV-LS4
|
tumor cells (CML), CD34+cells
|
NSVQSSW
|
|
AAV2
|
I-588
|
AAV-RGDLGLS
|
tumor cells
|
RGDLGLS
|
|
AAV2
|
I-588
|
AAV-RGDMSRE
|
tumor cells
|
RGDMSRE
|
|
AAV2
|
I-588
|
AAV-ESGLSQS
|
tumor cells
|
ESGLSQS
|
|
AAV2
|
I-588
|
AAV-EYRDSSG
|
tumor cells
|
EYRDSSG
|
|
AAV2
|
I-588
|
AAV-DLGSARA
|
tumor cells
|
DLGSARA
|
|
AAV2
|
I-588
|
AAV-GPQGKNS
|
tumor cells
|
GPQGKNS
|
|
AAV2
|
I-588
|
AAV-NSSRDLG
|
endothelial cells
|
NSSRDLG
|
|
AAV2
|
I-588
|
AAV-NDVRAVS
|
endothelial cells
|
NDVRAVS#
|
|
AAV2
|
I-588
|
AAV-PRSTSDP
|
lung (maybe endothelial cells)
|
PRSTSDP
|
|
AAV2
|
I-588
|
AAV-DIIRA
|
endothelial cells
|
DIIRA
|
|
AAV2
|
I-588
|
AAV-SYENV
|
endothelial cells
|
SYENVASRRPEG
|
|
AAV2
|
I-588
|
AAV-PENSV
|
endothelial cells
|
PENSVRRYGLEE
|
|
AAV2
|
I-588
|
AAV-LSLAS
|
endothelial cells
|
LSLASNRPTATS
|
|
AAV2
|
I-588
|
AAV-NDVWN
|
endothelial cells
|
NDVWNRDNSSKRGGTTEAS
|
|
AAV2
|
I-588
|
AAV-NRTYS
|
endothelial cells
|
NRTYSSTSNSTSRSEWDNS
|
|
AAV2
|
I-588
|
rAAV2-ESGHGYF
|
pulmonary endothelial cells
|
ESGHGYF
|
|
AAV2
|
I-588
|
AAV-GQHPRPG
|
cardiomyoblasts
|
GQHPRPG+
|
|
AAV2
|
I-588
|
AAV-PSVSPRP
|
cardiomyoblasts
|
PSVSPRP
|
|
AAV2
|
I-588
|
AAV2-VNSTRLP
|
cardiomyoblasts
|
VNSTRLP
|
|
AAV2
|
I-588
|
AAV-LSPVR
|
cardiomyoblasts
|
LSPVRPG
|
|
AAV2
|
I-588
|
AAV-MSSDP
|
cardiomyoblasts
|
MSSDPRRPPRDG
|
|
AAV2
|
I-588
|
AAV-GARPS
|
cardiomyoblasts
|
GARPSEVTTRPG
|
|
AAV2
|
I-588
|
AAV-GNEVL
|
cardiomyoblasts
|
GNEVLGTKPRAP
|
|
AAV2
|
I-588
|
AAV-KMRPG
|
cardiomyoblasts
|
KMRPGAMGTTGEGTRVTRE
|
|
AAV2
|
I-588
|
AAV588MTP
|
skeleton muscle
|
ASSLNIA
|
除了I-587和I-588外,I-453���、I-520&I-584、I-584��、I-585也已被成功用于開發(fā)新細(xì)胞靶向性工程化AAV(表2)�。
表2:非I-587和I-588衣殼變體
|
Serotype
|
Position
|
Name
|
Target Cell Type
|
Insert
|
|
AAV2
|
I-453
|
rRGD453ko
|
av integrin-positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-453
|
AAV-MNVRGDL
|
endothelial cells
|
MNVRGDL
|
|
AAV2
|
I-453
|
AAV-ENVRGDL
|
endothelial cells
|
ENVRGDL
|
|
AAV2
|
I-520 and I-584
|
A520/N584 (RGD)
|
av integrin-positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-584
|
A584-RGD4C
|
av integrin-positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-584
|
A584-RGD4CALS
|
av integrin-positive tumor cells
|
CDCRGDCFC
|
|
AAV2
|
I-585
|
AAV-?IV-NGR
|
CD13-positive tumor cells
|
NGRAHA
|
|
AAV2
|
I-585
|
AAV-PTP
|
Plectin-positive tumor cells
|
KTLLPTP
|
正如研究背景中所述����,體內(nèi)預(yù)存中和抗體是限制AAV廣泛臨床應(yīng)用的一大障礙��。血清學(xué)研究表明�����,大多數(shù)人都接觸過野生型AAV���,因此體內(nèi)可能已經(jīng)形成針對(duì)AAV的中和抗體。這些中和抗體可能會(huì)干擾AAV進(jìn)入靶細(xì)胞��、細(xì)胞內(nèi)運(yùn)輸和細(xì)胞核內(nèi)的解包裝����,從而阻止了轉(zhuǎn)導(dǎo)�����。據(jù)報(bào)道�����,抗AAV抗體在人群中的流行率約為40-80%�。其中,抗AAV2的中和抗體在人群中相對(duì)更流行�。為了克服這個(gè)障礙�,科學(xué)工作者以其他低免疫原性血清型為骨架�,并參考AAV2的衣殼修飾位點(diǎn),開發(fā)了一系列新的工程化AAV(表3)�。
表3:非AAV2骨架的工程化AAV
|
Serotype
|
Position
|
Name
|
Target Cell Type
|
Insert
|
|
AAV1
|
I-590
|
BAP-AAV1
|
Scavidin displaying BT4C (rat glioma)
|
GLNDIFEAQKIEWHE
|
|
AAV1
|
I-590
|
BAP-AAV1
|
endothelial cells
|
GLNDIFEAQKIEWHE plus CDCRGDCFC(RGD4C)
|
|
AAV1
|
I-590
|
AAV1-RGD
|
tumor cells, endothelial cells
|
CDCRGDCFC
|
|
AAV1
|
I-590
|
AAV1-RGD/BAP (90/10) (mosaic capsid)
|
tumor cells, endothelial cells
|
CDCRGDCFC and GLNDIFEAQKIEWHE
|
|
AAV1
|
I-590
|
Tet1c-AAV1 (mosaic capsid)
|
tetanus toxin GT1b receptor positive cells
|
HLNILSTLWKYR
|
|
AAV1
|
I-590a
|
AAV1.9-3-SKAGRSP
|
fibroblast
|
SKAGRSP
|
|
AAV5
|
I-575
|
BAP-AAV4
|
tumor cells
|
GLNDIFEAQKIEWHE
|
|
AAV6
|
I-585
|
AAV6-RGD
|
tumor cells
|
RGD
|
|
AAV6
|
I-585 plus Y705-731F+T492V
|
AAV6-RGD-Y705-731F+T492V
|
tumor cells
|
RGD
|
|
AAV6
|
I-585plus Y705-731F+T492V+K531E
|
AAV6-RGD-Y705-731F+T492V+K531E
|
tumor cells
|
RGD
|
|
AAV8
|
I-585c
|
AAV2/8-BP2
|
on-bipolar cells
|
PERTAMSLP
|
|
AAV8
|
I-590
|
AAV8-ESGLSOS
|
tumor cells
|
ESGLSOS135
|
|
AAV8
|
I-590
|
AAV8-ASSLNIA
|
heart (weakly improved transduction)
|
ASSLNIA122
|
|
AAV8
|
I-590d
|
AAV8-GQHPRPG
|
heart (weakly improved transduction)
|
GQHPRPG86
|
|
AAV8
|
I-590d
|
AAV8-SEGLKNL
|
liver
|
SEGLKNL
|
|
AAV9
|
I-589
|
AAV-SLRSPPS
|
endothelial cells, smooth muscle cells
|
SLRSPPS
|
|
AAV9
|
I-589
|
AAV-RGDLRVS
|
endothelial cells, smooth muscle cells
|
RGDLRVS
|
|
AAV9
|
I-589d
|
AAV9-NDVRAVS
|
endothelial cells
|
NDVRAVS82
|
|
AAV9
|
I-589d
|
AAV9-ESGLSOS
|
tumor cells (weak targeting)
|
ESGLSOS135
|
|
AAV9
|
I-588
|
AAV-PHP.B
|
CNS
|
TLAVPFK
|
|
AAV9
|
I-588
|
AAV-PHP.A
|
CNS
|
YTLSQGW
|
|
AAV9
|
I-588
|
AAV9-7m8
|
retinal cells
|
LGETTRP80
|
|
AAV9P1
|
not disclosed
|
AAV9P1
|
neuronal progenitor cells
|
RGDLGLS
|
如若了解靶向同一類細(xì)胞的所有血清型間的效果差異���,請參考Reference 2中對(duì)應(yīng)的參考文獻(xiàn)��。
|
維真提供快速即用工程化AAV——保證包裝成功!
|
|
您可以從表1中選擇您感興趣的AAV Cap質(zhì)粒���,我們將1-2周交付您即用型AAV病毒�����,并且承諾包裝不成功不收費(fèi)����,大大加快您的科研進(jìn)程!
此外�����,如果您對(duì)表2和表3中的質(zhì)粒感興趣�,我們也可以幫您進(jìn)行個(gè)性化定制,保證包裝成功�!
|
|
咨詢訂購:①撥打熱線電話400-077-2566; ②致電您的區(qū)域經(jīng)理����;③添加下方維真客服咨詢訂購!
|
|

|
5.結(jié)束語
目前AAV基因藥物的臨床研究大多數(shù)采用的是天然的衣殼�����,這些衣殼極易成為宿主酶的作用靶點(diǎn)��,從而影響它們的整體性能�����。此外��,天然AAV血清型具有組織親噬性�����,但通常趨向性比較廣泛��。因此�,必須使用高劑量的病毒才能達(dá)到治療效果���。而且�����,AAV載體的包裝容量和大多數(shù)人群中已存的中和抗體進(jìn)一步限制了AAV的臨床應(yīng)用���。為了克服這些限制,科學(xué)工作者將研究的重點(diǎn)聚焦在工程化AAV����,以進(jìn)一步提高其安全性����、有效性和特異性���,從而推動(dòng)基因治療領(lǐng)域的快速發(fā)展。在當(dāng)今基因治療的研究熱潮下�,為了加快您的科研進(jìn)程,我們建立了工程化AAV庫��,旨在為基因治療的發(fā)展貢獻(xiàn)自己的一份力���。
6.參考文獻(xiàn)
1. Lugin, M. L., et al. (2020). "Synthetically Engineered Adeno-Associated Virus for Efficient, Safe, and Versatile Gene Therapy Applications." ACS Nano 14(11): 14262-14283.
2. Buning, H. and A. Srivastava (2019). "Capsid Modifications for Targeting and Improving the Efficacy of AAV Vectors." Mol Ther Methods Clin Dev 12: 248-265.
3. Pipe, S., et al. (2019). "Clinical Considerations for Capsid Choice in the Development of Liver-Targeted AAV-Based Gene Transfer." Mol Ther Methods Clin Dev 15: 170-178.
4. Tseng, Y. S. and M. Agbandje-McKenna (2014). "Mapping the AAV Capsid Host Antibody Response toward the Development of Second Generation Gene Delivery Vectors." Front Immunol 5: 9.
5. Gurda, B. L., et al. (2013). "Capsid antibodies to different adeno-associated virus serotypes bind common regions." J Virol 87(16): 9111-9124.